Brain Computer Interfaces Are Now Giving Sight Back to the Blind
A 2mm chip restored sight more than 80% of blind trial patients, built by a former cofounder of Neuralink.

Source: garryslist.org
Source: garryslist.org
TL;DR
Science Corp’s PRIMA retinal implant restored vision in 81% of blind trial patients, published in the NEJM. Founder Max Hodak left Neuralink to build it, raised $230M, and the FDA is reviewing the application now.
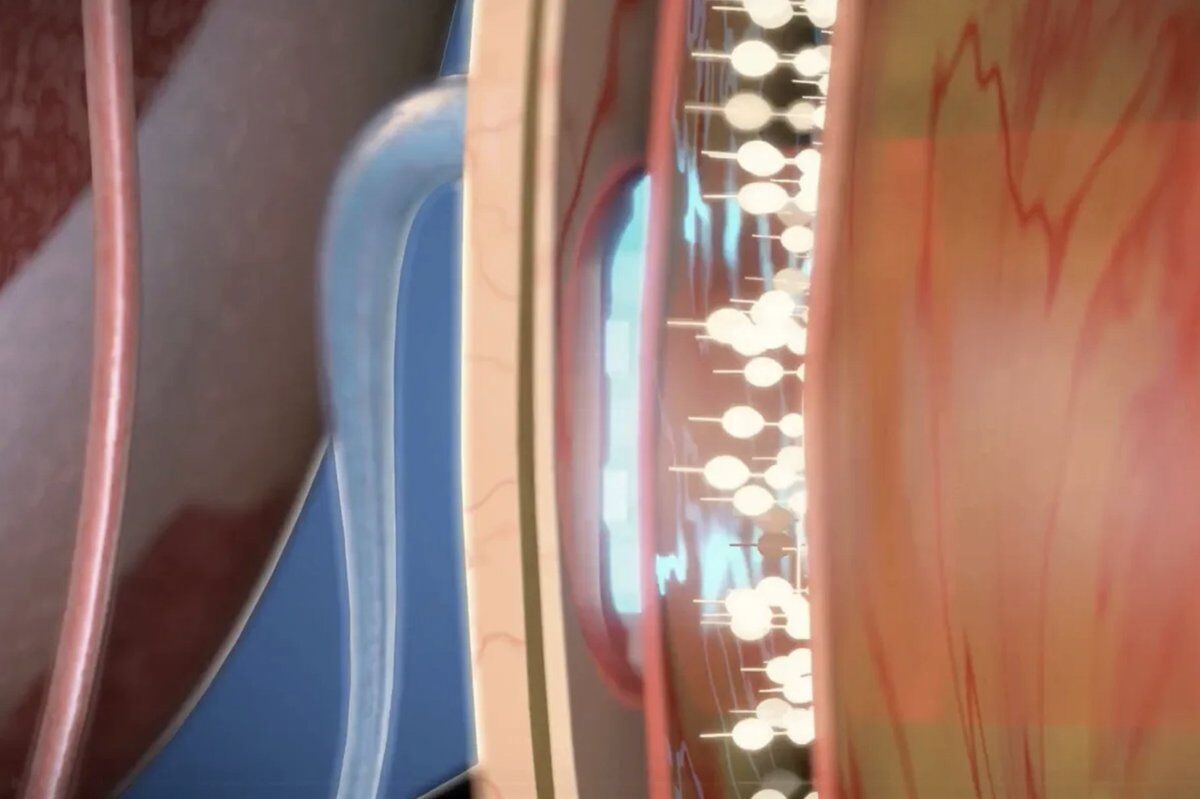
A chip smaller than your pinky nail. Thirty micrometers thin, half the thickness of a human hair. Surgeons slip it beneath the retina, where light-sensitive cells have been destroyed by age-related macular degeneration. Then the patient opens their eyes. Then they read.
26 of 32 blind patients regained clinically meaningful vision in a trial published in the New England Journal of Medicine. Average gain: five full lines on an eye chart. About 84% could read letters, numbers, and words again. This isn’t a press release. It’s peer-reviewed science.
Absolutely amazing. Vision restoration.
Congrats to @maxhodak_ and the @ScienceCorp_ team on their $230M Series C! Their PRIMA implant is letting blind people see again: improved vision in 26 of 32 trial patients with advanced macular degeneration. …
Science Corporation just raised $230M at a $1.5 billion valuation. The founder, Max Hodak, was formerly president of Neuralink. Y Combinator is an investor. The FDA is reviewing the application.
What 81% Actually Means
The PRIMAvera trial enrolled 38 participants with geographic atrophy from AMD, a leading cause of irreversible blindness. Of the 32 assessed at 12 months, 26 showed clinically meaningful improvement in visual acuity.
Six weren’t assessed: three had died (the study population skews elderly), one withdrew, two were unavailable. Accounting for missing data, researchers estimated 80% of all participants would have improved. Like any first experiment, there were adverse events: 26 serious ones across 19 of 38 participants. But 81% occurred within two months of surgery, and 95% of those resolved within two months. Natural peripheral vision was unaffected.
That’s Moorfields Eye Hospital’s senior vitreoretinal consultant. No other experimental treatment has ever restored central vision in patients with advanced geographic atrophy at this level of acuity.
‘Two Black Discs in My Eyes’
Sheila Irvine was a Moorfields patient in the trial. After the implant: prescription labels, tiny writing on tins, crossword puzzles. “It was dead exciting when I began seeing a letter,” she said. “The more hours I put in, the more I pick up.”
That’s what 81% means for one person. A woman doing a crossword for the first time in years.
How the Chip Works
The PRIMA system pairs a 2mm wireless implant under the retina with augmented-reality glasses and a pocket processor. The glasses capture a scene, AI identifies the main object, and near-infrared light projects the image onto the chip. The chip converts that light to electrical signals. Remaining retinal neurons relay them to the brain as vision.
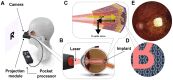
No battery inside the eye. No wires. The surgery takes under two hours, and any trained vitreoretinal surgeon can perform it. Built to scale to millions of patients, not stay locked in a lab.
From Neuralink to Patients First
Max Hodak co-founded Neuralink and served as its president. He left in 2021 to found Science Corp. Five years later: $490M raised, $1.5B valuation, the second-most valuable brain-computer interface company on earth.
He left the most famous BCI company to build one that would ship to patients first.

The $230M Series C includes Lightspeed, Khosla Ventures, Y Combinator, Quiet Capital, and IQT, the CIA’s venture arm for national security tech. Vinod Khosla thinks insurers will pay “hundreds of thousands of dollars” for the procedure. “He’s letting people see who have no hope of ever seeing again,” Khosla said. “That’s pretty transformative.”
Hodak sees one clear parallel: “We haven’t seen a new brain-computer interface business like this since the cochlear implant.” Cochlear implants are now a $2.3 billion market. Hundreds of thousands of people hear because of them. That’s the arc he’s betting on for vision.
Second Sight’s Warning
There’s a ghost in this field. Second Sight built the Argus II retinal prosthesis. CE mark in 2011. FDA approval in 2013. Over 350 patients implanted. Then the company went bankrupt and left them with dead implants in their eyes. Ross Doerr, an Argus II recipient: “Fantastic technology and a lousy company.” Second Sight stopped manufacturing and implanting Argus II in 2019
The current PRIMA treats the most advanced AMD cases, about 3,000 to 6,000 new US patients per year. AMD affects 5 million+ worldwide. That gap is the expansion path. Science Corp is launching a new trial for Stargardt disease and retinitis pigmentosa, inherited conditions that blind patients decades younger, with two patients targeted for implant by end of June. They’re building a higher-resolution chip for less severe vision loss, and a biohybrid brain device using engineered living neurons, with human testing by end of 2027. Neuralink’s Blindsight goes directly into the visual cortex, but they haven’t published data or announced trials. Science Corp may ship first.
European commercial launch is expected later this year. If it happens, PRIMA would become the first marketed BCI vision product in history.
Cochlear implants started as a niche curiosity. Now hundreds of thousands of people hear because of them. Sheila Irvine had two black discs where her eyes used to be. Now she does crossword puzzles. Five million more people are waiting. The FDA has the file on its desk.
Related Links
-
PRIMA Clinical Trial Results (NEJM via PubMed) (PubMed / NEJM)
-
Revolutionary implant allows patients with dry AMD to read again (Macular Society)
-
Second Sight's Argus II and the Rise of Obsolete Bionic Eyes (IEEE Spectrum)
Comments (0)
Sign in to join the conversation.